Index: Volume 5 Number 1
Next Article: Microplate PCR Kits
 |
By Anthony Salerno, Ph.D. PureBiotech LLC
For simple isolations and purifications of immunoglobulins such as; small-scale affinity purifications, immunoprecipitation reactions, single or automated immunoassays and bioassays, and separation of unbound labeled tracer.
-
Rapid, gentle & complete immobilization
- Easily automated
- No need for surfactants
- Will not clump or adhere to hydrophobic surfaces
- Economical purification of immunoglobulins
- Higher yields of concentrated target protein
PureBiotech LLC's Protein A particles are Magnetic Porous Glass (MPG®) particles to which protein A has been covalently attached. MPG® Protein A particles possess antibody binding properties similar to unconjugated protein A. The nominal binding capacity of 1 mg of particles is between 50-75 µg of immunoglobulin for strong species such as rabbit and human IgG1.
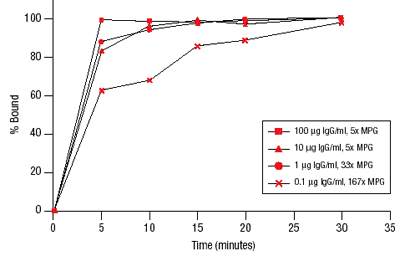
The high surface area and high loading of MPG® Protein A promote quick capture of target antibody. Using an excess of MPG® Protein A particles rabbit IgG can be cleared (>94%) within 5-10 minutes at concentrations as low as 1 µg/ml, as shown in Figure 1. Excess antibody present at mg/ml concentrations also saturates MPG® Protein A particles within 5 minutes.
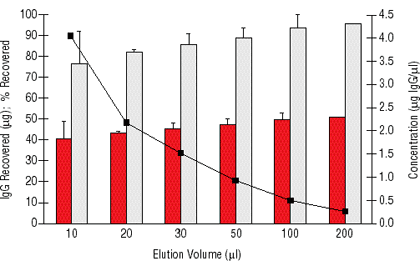
The high binding capacity of the particles combined with the ability to elute antibody in a minimal volume allows the recovery of highly concentrated target antibody/proteins. Figure 2 shows that more than 75% recovery of captured antibody can be achieved in as little as 10 µl volume while giving protein concentrations greater than 4 mg/ml. After neutralization, this allows for direct use of the supernatant in gel electrophoresis without the need for concentration or buffer exchange.
|
MPG® particles give higher yields of concentrated target protein than other available magnetic protein A particles. In addition to having higher binding capacity, magnetic porous glass particles do not clump. Polystyrene magnetic particles tend to clump and adhere to hydrophobic surfaces when surfactants or similar agents are eliminated from the buffers. This results in loss of particles to pipette tips, tube walls, etc. and increased elution volumes, which in turn lowers yield and dilutes recovered target protein (Table 1). MPG® Protein A performs well in high salt buffers without the presence of surfactants. Recovery can be essentially quantitative in as little as 50 µl of the appropriate buffer (Figure 2).
|
|
|||||||||||||||||||
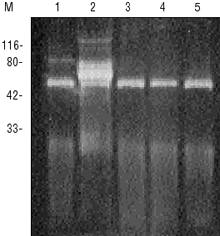
MPG® Protein A particles make mg scale magnetic batch purification of antibodies economically feasible. MPG® Protein A can be regenerated up to 12 times while still providing greater than 90% of initial binding capacity. The same high purity of antibody recovered from the particles during the first use is maintained in subsequent purifications. Only the heavy chain and diffuse light chain bands of polyclonal rabbit IgG are visable in the samples purified with MPG® Protein A (Figure 3). Regeneration is easy to do and takes only 5 minutes.
|
1 MPG® Protein A protocol (57.101)
Next Article: Microplate PCR Kits
© 2004-2023 PureBiotech LLC All rights reserved.