





Index: Volume 4 Number 1
Next Article: 3' Biotin End Labeling, made easy
 |
By Anthony J. Salerno, Ph.D., PureBiotech LLC
Introduction
A rapid method for the purification of high quality mRNA has been further optimized for speed and ease. This new method has been incorporated into PureBiotech LLC's solid phase affinity purification based "mRNA direct" kits which are noted for the ease and speed with which high quality mRNA can be prepared. PureBiotech, LLC's kits employ streptavidin coated magnetic porous glass (MPG®) as the solid phase combined with an oligo (dT)25 probe to isolate mRNA from either tissue, cells or total RNA. As shown in the report the ease and speed of these kits has been optimized while maintaining the high yield and purities of the resultant mRNA. This was accomplished by the inclusion of a prebound probe to the MPG® Streptavidin. Previously, the user needed to bind the probe to MPG® Streptavidin prior to the RNA isolation step. The PureBiotech LLC kits which incorporate this new improved prebound system are; MPG® Direct mRNA Purification Kit (MDRK), MPG® Guanidine Direct mRNA Purification Kit (MGRK), MPG® mRNA Purification Kit (MRRK), and SOLIDscript(tm) Solid Phase cDNA Synthesis Kit (SSP).
Speed & Ease
A study was done comparing the completed protocol times of the original mRNA kits to the newly optimized version. By including the new prebound MPG® Streptavidin-Oligo (dT)25 Complex and eliminating the binding and washing steps from the protocol a significant time savings was achieved. Table 1 below illustrates that the new completion time has been reduced by 25% in most instances.
Table 1: Time Comparison of Old Kit Format vs. New Kit Format
| mRNA Isolation Time | cDNA Generation Time | |||
| Kit Format | MDRK | MGRK | MRRK | SSP |
| Original | <20 min. | <20 min. | <20 min. | <110 min. |
| Improved | <15 min. | <15 min. | <15 min. | <104 min. |
Optimized parameters for easy mRNA isolation
In addition to improving the speed and ease of the kits, we focused on optimizing the conditions of the isolation. The first step was the optimization of the loading of biotinylated oligo (dT)25 to provide the highest mRNA yields from the new MPG® Streptavidin -oligo (dT)25 complex. The MPG® Streptavidin particles are independently quality control tested to meet a standard of at least 1500 pmol free biotin binding/mg of particles. High yields of mRNA are obtained over a fairly wide range of oligo (dT)25 loadings as shown in the histogram in Figure 1. It is apparent that loadings in excess of 800 pmol oligo (dT)25/mg MPG® result in little additional mRNA recovered. Therefore, the new kit format employs an optimal loading of 800 pmol oligo (dT)25/mg MPG® Streptavidin for maximal yield of mRNA.
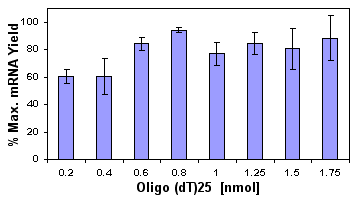 |
Figure 1: mRNA yeild as a function of oligo(dT)25 loading Using the PureBiotech LLC MDRK Kit the yield of mRNA recovered from 0.1 g of mouse liver was assayed spectrophotometrically. One mg MPG® Streptavidin and the indicated loadings of biotinylated oligo(dT)25 were used. |
Time Course for mRNA Binding to the MPG® Streptavidin- Oligo (dT)25 Complex
The length of time for complete binding was studied. Figure 2 shows the annealing kinetics for various samples. For all samples, the specific binding of polyadenylated mRNA to the MPG® Streptavidin-Oligo(dT)25 Complex was completed within 5 minutes. After 5 minutes, the majority of additional binding experienced is non-specific. As shown in Table 2, this weak non-specific binding of mRNA to the complex is easily removed in the subsequent washing steps. Table 2 also shows the amount of mRNA recovered after washing away impurities and loosely bound mRNA. This represents the maximum binding capacity or yield per mg MPG® Streptavidin- Oligo (dT)25 Complex. The lower binding capacity for globin compared to mouse liver RNA is consistent with the small size of globin mRNA [a and b < 600 bases1,2] relative to the length of the majority of mRNA present in mouse liver3.
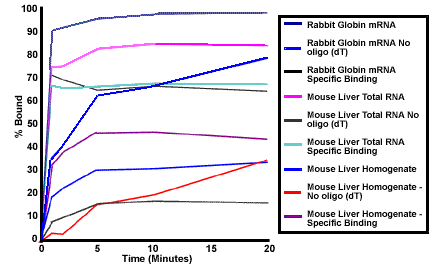
Figure 2: Binding Kinetics of Streptavidin-Oligo (dT)25 Complex
Rabbit globin RNA was 3' end-labeled with a-[32P] cordycepin triphosphate using yeast poly A polymerase and unincorporated label removed on a PureBiotech LLC SPIN-Pure™ Column (SCT25-50 ) followed by ethanol precipitation. The globin was mixed with mouse liver total RNA or mouse liver homogenate and used as a tracer to follow the binding and release of mRNA during purification using the MRRK and MGRK kit protocols, respectively. An excess of mRNA was used for each sample; 5 µg/0.4 ml for globin mRNA, 250 µg/0.7 ml for total RNA, and 1.5 ml of a 1/33 total dilution of mouse liver (homogenate). For the liver samples the globin tracer represented about 5% of the total mRNA present. MPG® Streptavidin with no bound oligo (dT)25 was used as a control to examine the nonspecific binding properties of mRNA. At various times, RNA samples were taken, mixed with MPG® Streptavidin, magnetically separated and the supernatant removed. The radioactivity remaining in the supernatant was assayed.
Table 2: Specific Binding Capacity per mg MPG® Streptavidin- Oligo (dT)25 Complex
| mRNA (µg) | |||
| Sample | Bound | Recovered | Retained on MPG® |
| Globin alone | 5.3 | 4.1 | < 0.1 |
| Globin + Mouse liver total RNA | 10.6a | 6.6 | < 0.1a |
| Globin + Mouse liver MGRK homogenate | 20.6a | 6.3 | < 0.1a |
| Globin alone without oligo (dT) | 0.7 | < 0.1 | < 0.1 |
| Globin + Mouse liver total RNA without oligo (dT) | 1.4ab | < 0.1 | < 0.1ab |
| Globin + Mouse liver MGRK homogenate without oligo (dT) | 8.9ac | < 0.1 | < 0.1ac |
bCalculated using the specific activity for mouse liver total RNA.
cCalculated using the specific activity for mouse liver MGRK Homogenate.
Shelf life of MPG® Streptavidin- Oligo (dT)25 Complex
Further studies were done to determine the shelf life of the new complex. The testing showed that the MPG® Streptavidin- Oligo (dT)25 Complex remains stable for at least one year when stored in the supplied buffer at 4¡C (Figure 3).
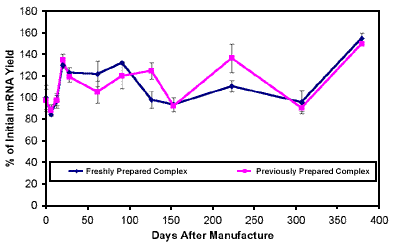 Figure 3. Storage Stability of MPG® Streptavidin-Oligo(dT)25 Complex. |
| On day 0 the MPG® Streptavidin- Oligo(dT)25 complex was prepared and assayed for mRNA binding capacity using excess mouse liver homogenate or total RNA. The complex was stored at 4°C in storage buffer and at the times indicated the complex was reassayed and compared to the binding capacity of freshly prepared complex using the same lot of MPG® Streptavidin. |
Purity of mRNA
Purity testing was done to confirm that the new complex yielded superior quality mRNA. Figure 4 compares the purity of mRNA isolated from a common total RNA sample using market leading cellulose and polystyrene based solid phase kits with PureBiotech, LLC's MPG® mRNA Purification Kit. Each solid phase was used according to the supplier's recommended protocol. The MPG® based kit yielded the purest mRNA. The others show substantial 18S rRNA contamination. This result reflects the highly effective and very time efficient washing achievable with MPG®.
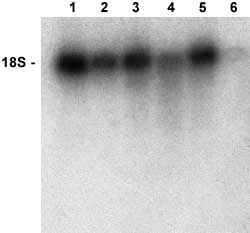 |
Figure 4. Purity comparison of mRNA isolated using market leading solid phase purification kits. Total RNA was prepared from mouse liver using PureBiotech, LLC's Total RNA Isolation Kit (MTRK). Messenger RNA was prepared from this RNA using the leading oligo (dT)- cellulose/spin basket based kit (lanes 1 and 2), the leading oligo (dT) polystyrene-latex/spin basket based kit (lanes 3 and 4), or PureBiotech, LLC's MPG mRNA Purification Kit (MRRK) (lanes 5 and 6). Each solid phase was used according to the supplier's recommended protocol. Samples (1 µg) of the non retained RNA (lanes 1,3, and 5) and purified mRNA (lanes 2,4 and 6)were electrophoresed on a 1% formaldehyde/agarose gel and a Northern blot was performed. The blot was hybridized to a 32P-labeled probe specific for 18S ribosomal RNA. |
Versatility and Convenience
The use of MPG® magnetic particle technology for direct mRNA isolation provides mRNA of high quality, yield, and purity now in an industry leading short time of 15 minutes or less. There is no longer any need for cumbersome manipulations. The purified mRNA can be eluted off the MPG® in any convenient volume of low salt elution buffer. The protocol is also readily adapted to microtiter plates for high throughput sample processing on robotic workstations.
- Heindell, H.C., Liu, A., Paddock, G.V., Studnicka, G.M. and Salser, W.A. (1978) Cell 15(1), 43.
- Kafatos, F.C., Efstratiadis, A., Forget, B.G. and Weissman, S.M. (1977) Proc. Natl. Acad. Sci. U.S.A. 74(12), 5618.
- Sambrook, J., Fritsch, E.F., and Maniatis, T. Molecular Cloning: A Laboratory Manual (1989) Cold Spring Harbor Laboratory Press, vol. 1, p 7.51.
Next Article: 3' Biotin End Labeling, made easy...
© 2004-2023 PureBiotech LLC All rights reserved.